ARIADNE
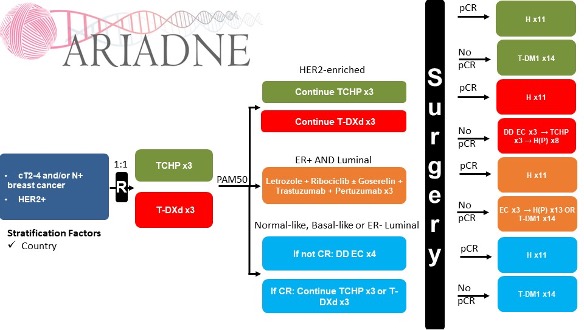
ARIADNE (NCT05900206) is an academic, international, open label, randomized, comparative phase IIB trial, conducted in five countries (Sweden, Norway, Italy, Belgium and Netherlands). Study co-PIs are Theodoros Foukakis and Alexios Matikas. A total of 370 patients will be enrolled (current inclusion, February 2026: 118). Patients with non-metastatic HER2-positive BC are randomized 1:1 and stratified per country to receive either i) standard neoadjuvant TCHP (docetaxel, carboplatin, trastuzumab, pertuzumab) for three cycles or ii) trastuzumab deruxtecan for three cycles. Further treatment is based on the intrinsic molecular subtype by Prosigna (PAM50) assay (baseline sample):
- HER2-enriched (approximately 65% of all patients): these patients continue with the same treatment for three more cycles.
- ER-positive and luminal (approximately 25% of all patients): these patients receive trastuzumab and pertuzumab for three cycles, combined with letrozole and ribociclib (plus goserelin for premenopausal patients) for two cycles
- ER-negative and luminal or basal-like or normal-like (approximately 10% of all patients): if the patient has attained radiologic complete response after three cycles according to contrast-enhanced MRI, she/he continues with the same previously allocated treatment for three more cycles. In case of no complete response, the patient receives four cycles of dose-dense epirubicin and cyclophosphamide (EC).
The primary endpoint of ARIADNE is locally assessed rate of pCR at the molecularly HER2-enriched population, defined as ypT0/Tis, ypN0, as determined at the surgical specimen by a pathologist blinded to treatment assignment (intention-to-treat analysis). Key secondary endpoints are: rates of complete radiologic response at three cycles by contrast-enhanced MRI; rates of pCR at the other two molecular groups and the two groups of the initial randomization; and event-free survival, defined as the time from randomization to disease progression, locoregional or distant recurrence, contralateral BC, or death due to any cause.
Research biopsies are taken at baseline, after three cycles of therapy in patients with radiologically residual cancer, and at surgery whereas blood samples are obtained at baseline, after cycle 1, cycle 3 and cycle 6, as well as annually at follow-up.
Relevant publications: