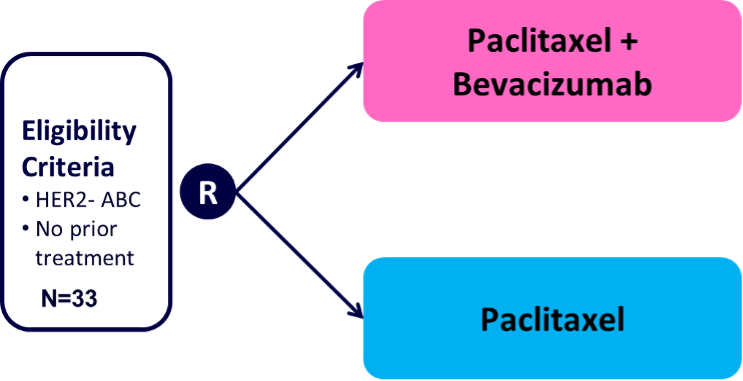
BEVPAC
BEVPAC (NCT01722968) is a randomized phase 2 trial that enrolled patients with advanced HER2-negative breast cancer that had not received prior therapy for metastatic disease. The study was initiated with a non-randomized, feasibility stage who were treated with bevacizumab and paclitaxel, in order to determine the safety of metastatic tumor biopsies during therapy with bevacizumab. In the second phase, patients were randomized (1:1) between two treatment arms: A. Bevacizumab + paclitaxel and B. Paclitaxel. Study PI is Theodoros Foukakis and 33 patients were enrolled.