PREDIX HER2
PREDIX HER2 (NCT02568839) is an academic, multicenter, phase 2, randomized clinical trial. Study PI is Thomas Hatschek. The trial was conducted 2014-2018 at 9 sites in Sweden and 202 patients were enrolled.
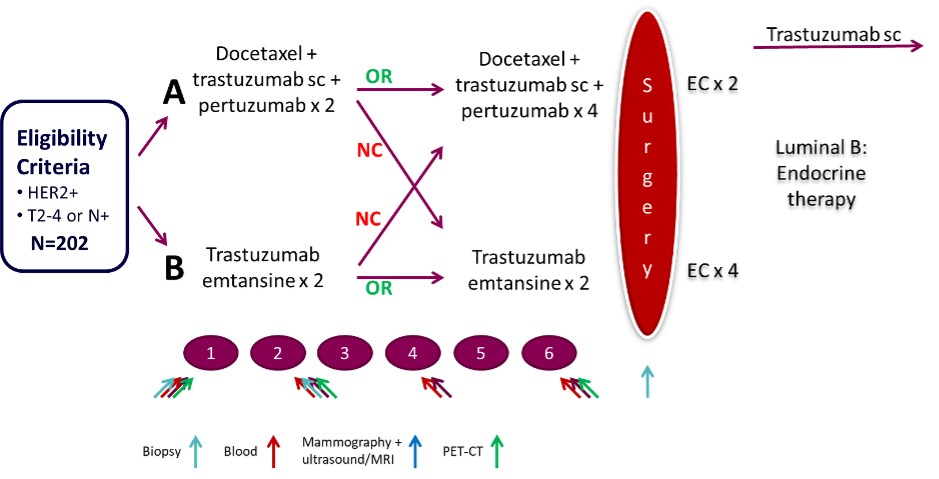
Eligible patients had a newly diagnosed HER2+ early breast cancer and were randomized to 6 three-weekly cycles of the combination of trastuzumab, pertuzumab and docetaxel, or trastuzumab-emtansine as neoadjuvant therapy. All patients received adjuvant anthracyclines. Patients who did not respond to treatment after 2 cycles switched to the other study arm. The primary endpoint of the trial was locally assessed pathologic complete response to treatment.
Blood samples and core tumor biopsies were obtained from all patients at baseline, after 2 cycles and at surgery. Response to treatment was monitored with radiological assessments and FDG-PET.
Relevant publications: