PREDIX Luminal B
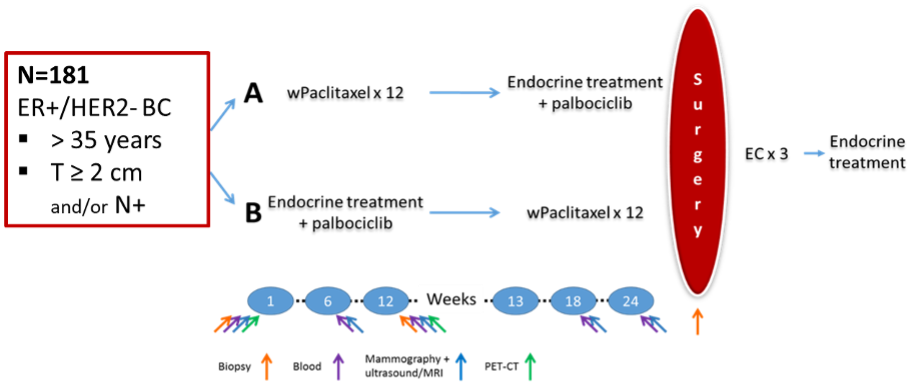
PREDIX Luminal B (NCT02603679) is an academic, multicenter, phase 2, randomized clinical trial. Study PI is Thomas Hatschek. The trial was conducted 2016-2021 at 3 sites in Stockholm and 181 patients were enrolled.
Eligible patients had locally advanced (primary tumor >2 cm and/or positive lymph nodes), ER-positive, HER2-negative breast cancer with an indication to receive neoadjuvant therapy. Treatment consisted of 12 weeks of either weekly paclitaxel or endocrine therapy in combination with palbociclib, or the reverse sequence. After surgery, all patients received 3 cycles of epirubicin and cyclophosphamide, followed by adjuvant endocrine therapy. The primary endpoint of the study was objective response after 12 weeks of treatment.
Biopsies were collected at baseline, at week 12 and at surgery, whereas PET/CT was performed at baseline and at week 12.
Relevant publications: