SBG 2004-1
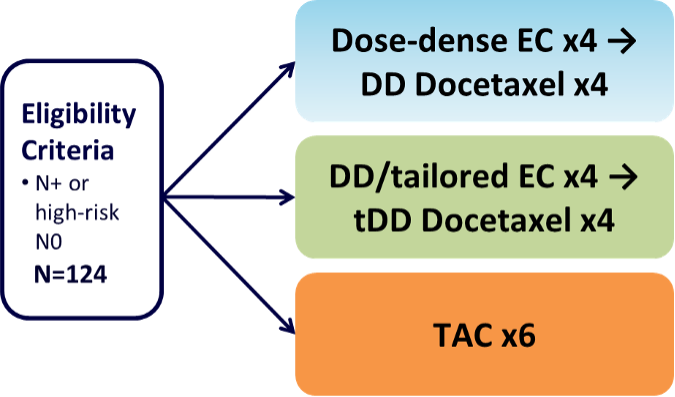
SBG 2004-1 is a randomized, multicenter phase II trial which compared three different regimens as adjuvant treatment for completely resected breast cancer. The study was conducted at 10 collaborative centers of the Swedish Breast Group in Sweden and enrolled 124 patients. Study PI is Jonas Bergh. Ten-year follow-up has been collected (end of study).
Patients with node positive (at least 4 positive nodes if ER+, 1 positive node if ER-) were randomly assigned to one of three treatment groups: tailored epirubicin and cyclophosphamide every 2 weeks followed by tailored docetaxel every 2 weeks, each for 4 cycles; fixed dose epirubicin and cyclophosphamide every 2 weeks followed by fixed dose docetaxel every 2 weeks, each administered for 4 cycles; or six cycles of the TAC regimen administered every 3 weeks.
Relevant publications: